DocIDManager
v1.0.0 — Dagon Release
A purpose-built desktop application for document identification in GxP-regulated organisations. Consistent, traceable, audit-ready — from day one.
The document register problem most organisations have and few address properly.
Most GxP organisations manage their document numbering the same way: a spreadsheet, maintained manually, version-controlled by filename, and quietly non-compliant. It works until it doesn't: when it fails, it fails during an inspection.
DocIDManager replaces ad hoc numbering with a controlled, auditable system. It runs entirely on Windows, requires no server, no cloud connectivity, and no ongoing IT infrastructure. Data stays on your organisation's systems. The application generates standardised document ID numbers automatically, according to a defined schema, and records every assignment in a tamper-evident audit trail, so every entry is traceable, every gap is visible, and no inspector can question the chronology.
It is designed for Quality Assurance and Regulatory Affairs teams in pharmaceutical, biotechnology, and medical device organisations. Licensed per site under an annual subscription, with no per-seat fees within that site.
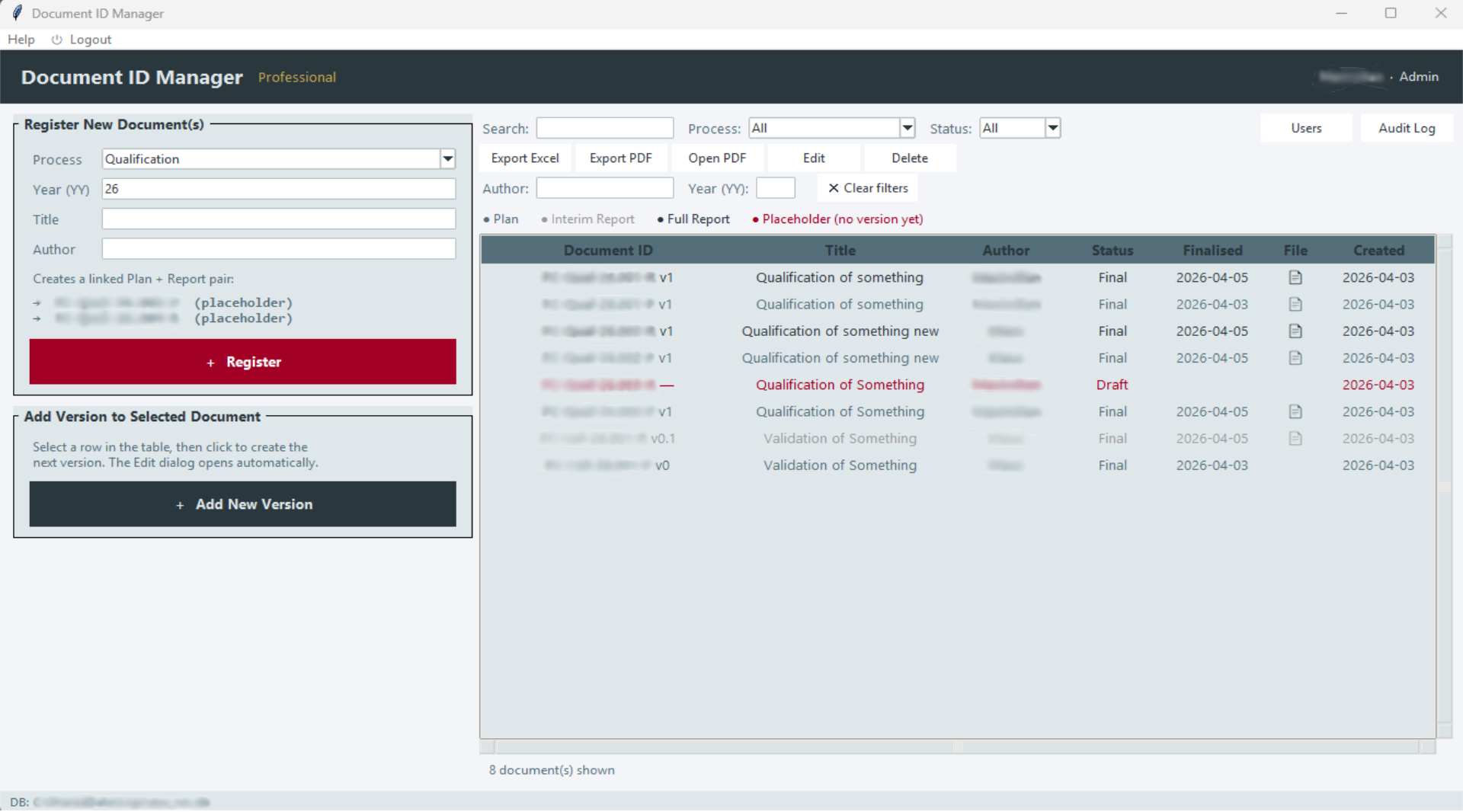
DocIDManager · document register view · Windows desktop application
Capabilities & Regulatory Alignment
At its core, the application generates sequential, schema-conformant document ID numbers, automatically, without manual entry, and without the numbering conflicts or duplicates that spreadsheet systems routinely produce. The Professional tier extends this with a SHA-256 cryptographic hash chain: a tamper-evident log in which every entry is linked to the previous, making retroactive modification detectable.
When a licence expires, the application transitions to read-only mode rather than shutting down. Existing records and the audit trail remain fully accessible — important in GMP environments where data retention obligations outlast any software subscription.
The tool has been developed to support working in a regulated under applicable FDA, EMA, ISO, and ICH requirements. Electronic signatures are explicitly out of scope. Standard and Professional licences include a pre-authored IQ/OQ validation template and Software Description Document, reducing the qualification effort required on your side.
Who Uses It
QA managers use it to establish a standardised ID schema across their organisation and enforce it systematically, removing the reliance on individual memory or manual conventions that tend to collapse under inspection pressure. Regulatory Affairs teams rely on it to ensure every document entering a submission carries a traceable, consistent identifier, reducing the back-and-forth with CMC and review teams that inconsistent numbering reliably causes.
For Document Control teams, it replaces the master numbering spreadsheet with a controlled, auditable database: one that cannot be accidentally overwritten, misconfigured, or corrupted by the person who happened to have the file open last. For biotech start-ups, it solves the common failure mode of retroactively restructuring document numbering ahead of a first FDA or EMA inspection. Starting right costs significantly less than correcting course under regulatory scrutiny.
Annual Site Licences
All tiers are priced as annual site licences — unlimited users within the licensed site, no per-seat fees. Pricing applies per single company site.
Entry
Basic
€990
per year · site licence
- Automated document ID generation
- Local SQLite database
- Read-only mode on licence expiry
- Email support
Recommended
Standard
€2,490
per year · site licence
- Everything in Basic
- IQ / OQ Validation Package
- Software Description Document
- Priority support & SLA
Full Compliance
Professional
€3,990
per year · site licence
- Everything in Standard
- SHA-256 cryptographic audit trail
- Field-level audit log
- Mandatory reason-for-change
Qualification support, pre-authored.
Standard and Professional licences ship with a complete set of GxP-aligned documentation. The framework is done — your team executes, reviews, and signs off.
Ready to establish compliant document control?
Contact SciReg Consult to discuss your requirements, request a demonstration, or arrange a licence for your organisation.